
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | ICMAB-CSIC Viñas Teixidor | + 3067 word(s) | 3067 | 2021-12-23 07:06:32 |
Video Upload Options
Glioblastoma multiforme (GBM) is one of the most common malignant brain tumors. Although a variety of GBMs is initially susceptible to chemotherapy, the development of multi-drug resistance and recurrence is frequent.
1. Introduction
Glioblastoma multiforme (GBM) is one of the most common malignant brain tumors among adults and the second most common cause of cancer-related deaths in children under the age of 15[1]. It has a poor prognosis, and the life expectancy of patients is approximately one year[2]. Even with treatment, almost all GBMs develop resistance to therapy and recurrence[3][4][5]. Therefore, there is an urgent need for more efficient treatment modalities for GBM. In that context, boron neutron capture therapy (BNCT) has emerged as a therapeutic strategy that aims at selectively destroying tumors by targeting tumor cells with 10B-containing compounds, followed by deposition of high doses of radiation on those cells through a thermal neutron capture reaction to yield high linear energy transfer (high-LET) α particles and recoiling 7Li nuclei [6]. Since the path’s length of the α-particle is approximately 9–10 μm, similar to a single cell diameter, it is possible to selectively irradiate a tumor cell with a large dose of radiation. In theory, the labeled cells will die, whereas the unlabeled ones will remain undamaged even if located in close vicinity to the target cells.
2. Cell Viability Assays
The in vitro assessment of the inhibitory capacity of [o-COSAN]− and [8,8′-I2-o-COSAN]− was performed by evaluating cell viability by means of the MTT assay. The experiments were carried out by adding increasing amounts of [o-COSAN]− or [8,8′-I2-o-COSAN]− to the incubation medium containing the cells (U87 and T98G, respectively) for 24 h in normoxic condition (at 37 °C and 5% CO2 in a cell incubator). For both cell lines, [8,8′-I2-o-COSAN]− was found to be more toxic than [o-COSAN]−. This has already been reported in previous papers for other cell lines[7] and is presumably due to the greater ability of [8,8′-I2-o-COSAN]− in crossing biological membranes compared to [o-COSAN]−[8][9]. The IC50 values of the two different compounds for both cell lines determined for different incubation periods are reported. [o-COSAN]− is not cytotoxic up to 48 h of incubation in the U87 cells. However, at 48 h incubation, this compound is more active in the T98G cells. At this incubation time, the corresponding iodinated compound seemed to be even more active in both cell lines.
Results on the cytotoxic behavior of these compounds were surprising, taking into consideration other reports in the literature on the characteristics of these glioblastoma (GMB) cells. In fact, in contrast with U87 cells, the T98G cells are reported as chemoresistant (Temozolomide/TMZ) GMB cells[10]. Therefore, our results suggest that these compounds, in particular the [8,8′-I2-o-COSAN]− are worth further investigating either alone or in combination with other conventional chemotherapeutics, in a larger panel of chemo-resistant GBM cell lines.
3. Effect of Cobaltallabis (Dicarbollides) in GBM Spheroids’ Viability
The spheroids were exposed to the two metallabis(dicarbollides) on the third day of culture in three different dosages. The concentrations of each compound were chosen based on the IC50 values previously determined for the monolayer culture of this cell line at 72 h, using the MTT assay, and then using a lower and a higher concentration than those IC50 values (0.5 × IC50 and 1.25 − 5 × IC50). After 72 h of exposure to the compounds (6th day of spheroid culture), the viability of the U87 and T98G spheroids was assessed by the acid phosphatase (APH) assay. Physical characterization of the spheroids upon incubation with the compounds, which included the measurement of the diameter, area, and circularity, was also performed daily, from day 3 to day 6 of culture.
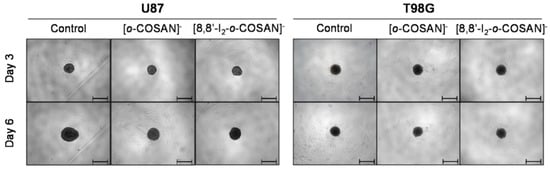
Figure 1. Representative images of the U87 and T98G spheroids before (day 3 of culture) and after 72 h (day 6 of culture) of exposure to the two compounds at their IC50 concentration. Controls consist of spheroids incubated only with medium. Scale bars correspond to 500 μm.
In a qualitative analysis, the administration of the compounds did not seem to significantly affect the shape and integrity of the spheroids. Quantitatively, concerning the growth and size of the spheroids, both compounds affected the growth of the U87 spheroids, with a statistically significant decrease being observed in the spheroids’ area at day 3 of treatment for all the three concentrations tested: the IC50 (Figure 2A), above IC50, and even below IC50 concentrations. In contrast, for the T98G cell line, no changes were detected in the growth of the spheroids for any of the concentrations tested (Figure 2C).
Figure 2. Effects of incubation of [o-COSAN]− and [8,8′-I2-o-COSAN]− on U87 (top panels) and T98G (bottom panels) spheroids. (A) U87 and (C) T98G spheroids growth after incubation with IC50 concentrations of [o-COSAN]− and [8,8′-I2-o-COSAN]−, represented by the mean spheroids area (in µM2) as a function of the number of days in culture, and (B) and (D) cellular viability (%) at 72 h, assessed by the APH assay, in parallel to monolayer cultured cells. Controls consist of spheroids or monolayer cultured cells incubated only with medium. Data are presented as the average ± SEM of 3 independent assays. Statistical significance was calculated using one-way ANOVA, followed by Dunnett’s test comparing treated spheroids/cells with control spheroids/cells (* p ≤ 0.05, ** p ≤ 0.01, *** p ≤ 0.001, and **** p ≤ 0.0001).Overall, the circularity of the spheroids was not severely affected by the administration of the compounds, maintaining, in most cases, a regular shape, which is reflected by circularity values very close to 1.
The viability of the spheroids after incubation with the metallabis(dicarbollides) for 72 h was determined using the APH assay and was performed in parallel also for monolayer-cultured cells. The viability results of the U87 and T98G spheroids for the compounds with all the concentrations tested are represented in Figure 2B,D, respectively. In general, these results reflect the growth behavior observed for the spheroids. There was an obvious decrease in the viability of the spheroids from U87 treated with both compounds. However, using the concentration corresponding to the IC50 previously determined in monolayer-cultured cells by the MTT assay, no compound led to a statistically significant decrease in the spheroids’ viability from both cell lines (Figure 2B,D). When using a higher concentration (Figure 2B), only spheroids from the U87 cell line were found to have reduced viability after incubation with both compounds, suggesting an increased resistance from T98G spheroids, contrarily to the observation in the monolayer culture.
The same assay was performed in parallel for monolayer-cultured cells, and a dose-dependent decrease in viability was observed in those conditions (see Figure 2B,D). When compared with 3D spheroids, both U87 and T98G cells grown as monolayer cultures were more sensitive to the compounds tested. In a way, the results reflected the disparity between 3D and monolayer cultures as 3D culture systems are frequently more refractory to anti-cancer treatments due to limited drug penetration and activation of several resistance mechanisms[11]. In fact, spheroid models can mimic the metabolic and proliferative gradients of in vivo tumors, with consequent changes in cellular phenotype and status, exhibiting multicellular chemoresistance.4. In Vivo Tests with Caenorhabditis elegans (C. elegans)
After performing in vitro testing of Na[o-COSAN] and Na[8,8′-I2-o-COSAN], we evaluated their toxicity in vivo using the small invertebrate C. elegans, which complement previous results and offer a physiologically relevant environment. C. elegans is a free-living nematode that, due to its simplicity, easiness of growth in large populations, and transparency, is used to assess the toxicity of drugs and materials[12][13] (Figure 3a). Despite these advantages, we acknowledge that C. elegans is evolutionarily distant from humans, and it lacks some defined organs/tissues, including blood, a brain, and the ability to develop a tumor. However, the use of this worm in material science offers the possibility to screen different nanomaterials and drugs before going one step further, reducing time, cost, and experiments with complex animal models and therefore fulfilling the 3Rs rule (replacement, reduction, and refinement). With this information in hand, further evaluation in complex animal models could next be performed.
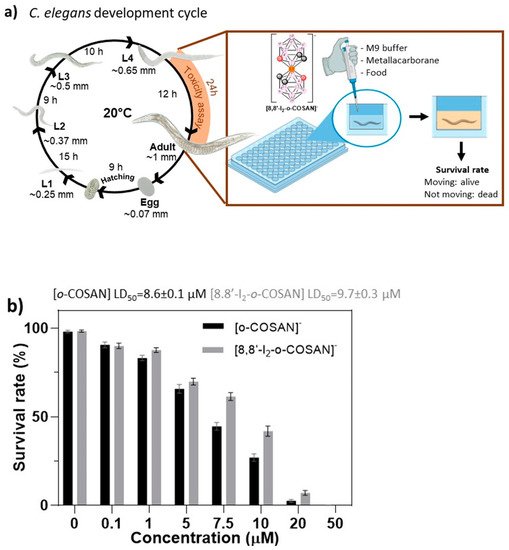
Figure 3. Toxicological evaluation of Na[o-COSAN] and Na[8,8′-I2-o-COSAN]. (a) Schematic representation of C. elegans’ life cycle at 20 °C and exposure of the components from L4 stage to adult stage. (b) Survival rate of C. elegans after 24 h of exposure towards concentrations in a range 0–50 µM of Na[o-COSAN] (black) and Na[8,8′-I2-o-COSAN] (grey).
Synchronized L4 C. elegans were exposed to Na[o-COSAN] and Na[8,8′-I2-o-COSAN] at a concentration ranging from 0 to 50 µM for 24 h of incubation. Toxicity is easily screened by the survival of the worms (existence of movement)[12], facilitating the determination of the lethal dose 50% (LD50). LD50 for Na[o-COSAN] and Na[8,8′-I2-o-COSAN] were 8.6 ± 0.1 µM and 9.7 ± 0.3 µM, respectively; those values are close to the IC50 determined for T98G in vitro after 72 h (Figure 3b). The small anionic [o-COSAN]− moieties, as described previously[14][15][16], could interact with the amine groups in the plethora of biomolecules of the organism and contribute to the toxicity.
The presence of both cobaltabis(dicarbollides) in the medium stop the development of worms at the L4 stage, whereas control worms could reach adulthood. Moreover, when worms were exposed to high concentrations of compounds (200 µM), a change towards yellowish color was observed, indicating accumulation in the body and the availability to cross C. elegans biological membranes in vivo.
Recently, the cell-cycle process of two glioma initiating cells (GICs: proneural GIC7 and mesenchymal PG88) treated with 200 µM Na[o-COSAN] 5 h was analyzed by flow cytometry[17]. When cells were recovered, 43 h after treatment, no differences were observed concerning control cells, neither GIC7 nor PG88. They might enter in the G2/M phase to complete cellular division, indicating that Na[o-COSAN] has a cytostatic effect in both GICs cells’ lines. Consequently, after the study with synchronized L4 C. elegans’ worms, we used C. elegans’ embryos to evaluate if the small cobaltabis(dicarbollide) anions would be able to interact with multilayer eggshell, crossing it and interfering with the embryonic development. C. elegans’ embryos are protected with a resistant and complex peri-embryonic shell of 300–400 nm thick[18], structured with different layers (vitelline, chitin, chondroitin, and extra-embryonic), whose chemical composition is polysaccharides, proteins, and lipids[18].
It was observed that C. elegans’ embryos did not develop to larvae stage after being treated with 200 µM in an M9 buffer solution of each compound (Na[o-COSAN] and Na[8,8′-I2-o-COSAN]) for 24 h at RT. This fact indicates that the compounds inhibit the development of embryos probably because of their interaction with the components of the protective shell, either at the peri-embryonic shell or at the cytoplasm.
Trying to elucidate on the interaction type along with on where the small anions ([o-COSAN]− and [8,8′-I2-o-COSAN]−) locate, a thorough investigation study of the eggs was designed and performed. The extraction of different fractions at different steps and the analysis using different chemo-physical techniques such as optical and SEM microscopies, ATR-IR, UV-vis, EDX, and RMN spectroscopies allowed the identification of the COSAN moieties at the different structures.
Firstly, optical microscopy images of the treated eggs (after washing three times with M9) clearly indicated a change in the color (Figure 4a), which would agree with the presence of the small anions in the eggs. Then, the ATR-FTIR spectrum of the dry eggs’ samples (eggs-[o-COSAN]− and eggs-[8,8′-I2-o-COSAN]−, respectively) was run. The spectra displayed the characteristic B-H band around 2.550 cm−1 and a change in the fingerprint area (1233–400 cm−1 range) confirming the metallacarborane’s presence in the treated eggs and their interaction with eggs’ components[10]. EDX measurements of the dried eggs-[o-COSAN]− and eggs-[8,8′-I2-o-COSAN]− samples confirm the presence of cobalt in both samples as well as iodine in the eggs-[8,8′-I2-o-COSAN]− (Figure 4).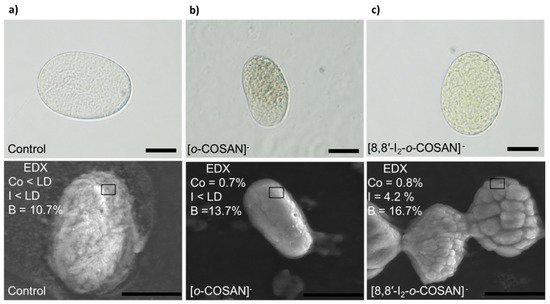
Figure 4. Optical and SEM images of the C. elegans embryos. (a) eggs-control; (b) after treatment with 200 μM of Na[o-COSAN] for 24 h at room temperature under gentle agitation; (c) after treatment with 200 μM of Na[8,8′-I2-o-COSAN] for 24 h at room temperature under gentle agitation. Bar size 20 μm.
The UV-vis spectra of the extracted aqueous solutions also confirmed the presence of the anionic cluster. However, the residual pellets still displayed a clear yellow color, which may indicate that the metallacarboranes remained in both the treated embryos after aqueous extraction. The fact that the metallabis (dicarbollides) remained in the dried pellets was confirmed by ATR-IR spectra, which clearly displayed the band around 2550 cm−1 corresponding to the ν(B-H). Taking into account that the sodium salts of the small anions, [o-COSAN]− and [8,8′-I2-o-COSAN]−, are water-soluble (1509 and 210 mM, respectively)[19], the former unsuccessful extraction with D2O suggests that the small anionic molecules strongly interact with amino-containing biomolecules in the embryos. It is known that these anions have the capacity to strongly interact with DNA[20][21], aminoacids[14][15][16] and proteins[22][23][24] through hydrogen or dihydrogen and halogen bonds (Cc-H···O and Cc-H···H-B or N-H···H-B or B-I···H-N, respectively). The impact of halogen (F, Cl, Br, and I) insertion on the biological activity and isozyme selectivity have been reported to be a source of inspiration for medicinal chemists[25][26][27][28].
Therefore, with the aim to extract the retained cobaltabis(dicarbollides) from the respective eggs’ pellets, d6-acetone was added to these pellets that lost the yellow color after the acetone extraction. The yellowish d6-acetone solution under 1H{11B} and 11B{1H} NMR spectroscopy unambiguously confirmed the presence of the cobaltabis(dicarbollide) in the samples. As an example, the 1H{11B} spectrum of the treated embryos with Na[8,8′-I2-o-COSAN] displays a broad resonance at 4.41 ppm that corresponds to Cc-H as well as the signals at 3.24, 3.07, 2.61, and 1.84 ppm corresponding to B-H resonances, while its 11B{1H} spectrum fully overlaps with the one of the parent Na[8,8′-I2-o-COSAN]. The ATR-IR spectra of the dried yellowish d6-acetone solution displayed the typical B-H band around 2.550 cm−1, which undoubtedly agrees with the presence of the anionic small metallacarborane anions in the dry eggs’ pellets before d6-acetone extraction.
In conclusion, Na[o-COSAN] and Na[8,8′-I2-o-COSAN] compounds were located in the C. elegans’ embryos after being treated with a 200 µM solution of the compounds for 24 h at RT under gentle agitation. The small cobaltacarborane anions form hybrids (eggs-[o-COSAN]− and eggs-[8,8′-I2-o-COSAN]−) with the chemical components of the embryos providing a yellowish color to the eggs. The hybrids are lightly soluble in water but were extracted with acetone. The C. elegans eggs’ development was affected by the hybrids’ formation between either Na[o-COSAN] or Na[8,8′-I2-o-COSAN] and the embryos’ chemical composition, be it DNA or proteins.
5. Cell Uptake Experiments
In order to assess whether the amount of boron taken up by target cells was enough to allow the set-up of an efficient BNCT procedure, U87 and T98G cells were incubated with increasing concentrations of [o-COSAN]− and [8,8′-I2-o-COSAN]−, were analyzed for their boron content by ICP-MS technique. Both anions, [o-COSAN]− and [8,8′-I2-o-COSAN]−, contain natural abundant boron; thus, the amount of 10B internalized was calculated as 19.9% of the total boron. Figure 5 shows that [o-COSAN]− concentrations of 10 μM and 20 μM for U87 and T98G, respectively, in the culture medium were sufficient to load U87 with 2.7 and 12.9 μg/g of 10B and T98G with 11.8 and 18.5 μg/g of 10B. [8,8′-I2-o-COSAN]− concentrations of 10 μM and 20 μM led to a higher loading for both cell lines, specifically of 29.5 and 49.8 μg/g of 10B for U87 and 53.6 and 69.7 μg/g of 10B for T98G. These quantities are well above the minimum boron threshold needed to perform BNCT. Under these experimental conditions, the cell viability measured by MTT was above 75% for all the cell lines.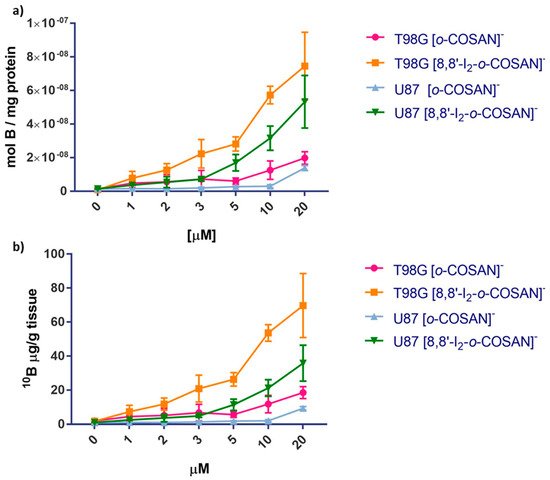
Figure 10. Upload of boron in U87 and T98G cells incubated in the presence of increasing concentration of [o-COSAN]− and [I2-o-COSAN]− for 24 h at 37 °C expressed as mol B/mg of protein (a) and µg 10B/g tissue (b). Error bars indicate the SD.
From these results, researchers can conclude that the T98G cells, resistant to conventional radiotherapy, internalize significantly higher amounts of 10B with respect to U87 cells. This gives more importance to the use of BNCT as an alternative strategy for the treatment of this type of glioblastoma tumor.
6. BNCT Studies in Glioblastoma Cells
The viability of cells irradiated with thermal neutron treated ([o-COSAN]−-IRR and [8,8′-I2-o-COSAN]−-IRR) and not treated (CTRL-IRR) with Na[o-COSAN] and Na[8,8′-I2-o-COSAN] was evaluated and compared with the corresponding non-irradiated ones (i.e., CTRL, [o-COSAN]− and [8,8′-I2-o-COSAN]−, respectively). Before irradiation, Na[o-COSAN] and Na[8,8′-I2-o-COSAN] 20 µM were incubated for 24 h at 37 °C with U87 and T98G cells. The irradiation time and reactor power were 15′ and 30 kW, respectively. Figure 6 shows the percentage of cells that survived neutron irradiation with respect to the non-irradiated control cells. From these results, it was evident that the T98G cells were more resistant to neutron irradiation, as no apparent cell-killing effect was observed for untreated cells. In contrast with Na[o-COSAN], the viability of T98G cells treated with Na[8,8′-I2-o-COSAN] was significantly reduced upon the irradiation treatment (Figure 6). As expected, the more pronounced effect of neutron irradiation was observed on these cells for the iodinated compound as a consequence of the higher amount of boron internalized (Figure 5). The U87 cells showed to be more sensitive to irradiation. In fact, a decrease of ca. 30% in the cell viability after irradiation was observed for untreated cells. For these cells, the trend was different; the Na[o-COSAN] presented a more promising profile for BNCT due to the cytotoxic behavior of its iodinated analog in these U87 cells.
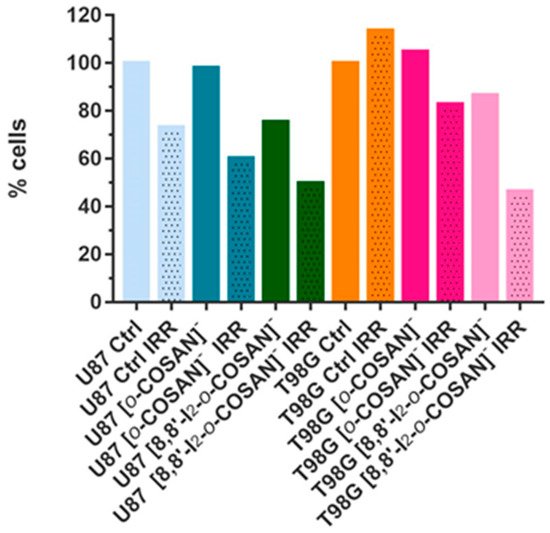
Figure 6. Percentage of viable U87 and T98G cells 24 h after neutron irradiation.
Results showed that the T98G cells seemed to be more resistant to neutron irradiation, which is somehow in agreement with other previous reports[10][29].
To sum up, these studies unveil the potential of this type of molecules to be further evaluated as BNCT agents and may have a relevant role in eliminating glioblastoma cells resistant to other types of radiotherapies.References
- Fitzmaurice, C.; Dicker, D.; Pain, A.; Hamavid, H.; Moradi-Lakeh, M.; MacIntyre, M.F.; Allen, C.; Hansen, G.; Woodbrook, R.; Wolfe, C.; et al. The Global Burden of cancer 2013. JAMA Oncol. 2015, 1, 505.
- Ohka, F.; Natsume, A.; Wakabayashi, T. Current trends in targeted therapies for glioblastoma multiforme. Neurol. Res. Int. 2012, 2012, 878425, doi:10.1155/2012/878425.
- Goellner, E.M.; Grimme, B.; Brown, A.R.; Lin, Y.-C.; Wang, X.-H.; Sugrue, K.F.; Mitchell, L.; Trivedi, R.N.; Tang, J.; Sobol, R.W. Overcoming temozolomide resistance in glioblastoma via dual inhibition of NAD+ biosynthesis and base excision repair. Cancer Res. 2011, 71, 2308–2317, doi:10.1158/0008-5472.CAN-10-3213.
- Moreno, M.; Pedrosa, L.; Paré, L.; Pineda, E.; Bejarano, L.; Martínez, J.; Balasubramaniyan, V.; Ezhilarasan, R.; Kallarackal, N.; Kim, S.-H.; et al. GPR56/ADGRG1 inhibits mesenchymal differentiation and radioresistance in glioblastoma. Cell Rep. 2017, 21, 2183–2197, https://doi.org/10.1016/j.celrep.2017.10.083.
- Stupp, R.; Mason, W.P.; van den Bent, M.J.; Weller, M.; Fisher, B.; Taphoorn, M.J.B.; Belanger, K.; Brandes, A.A.; Marosi, C.; Bogdahn, U.; et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N. Engl. J. Med. 2005, 352, 987–996, doi:10.1056/NEJMoa043330.
- Nedunchezhian, K.; Aswath, N.; Thiruppathy, M.; Thirugnanamurthy, S. Boron neutron capture therapy—A literature review. J. Clin. Diagn. Res. 2016, 10, ZE01–ZE04.
- Navascuez, M.; Dupin, D.; Grand, H.-J.; Gómez-Vallejo, V.; Loinaz, I.; Cosido, U.; Llop, J. COSAN-stabilised omega-3 oil-in-water nanoemulsions to prolong lung residence time for poorly water soluble drugs. Chem. Commun. 2020, 56, 8972–8975.
- Tarrés, M.; Canetta, E.; Viñas, C.; Teixidor, F.; Harwood, A.J. Imaging in living cells using νB–H Raman spectroscopy: Monitoring COSAN uptake. Chem. Commun. 2014, 50, 3370–3372.
- Verdiá-Báguena, C.; Alcaraz, A.; Aguilella, V.M.; Cioran, A.M.; Tachikawa, S.; Nakamura, H.; Teixidor, F.; Viñas, C. Amphiphilic COSAN and I2-COSAN crossing synthetic lipid membranes: Planar bilayers and liposomes. Chem. Commun. 2014, 50, 6700–6703, doi:10.1039/C4CC01283F.
- Lee, S.Y. Temozolomide resistance in glioblastoma multiforme. Genes Dis. 2016, 11, 198–210, doi:10.1016/j.gendis.2016.04.007.
- Mittler, F.; Obeïd, P.; Rulina, A.V.; Haguet, V.; Gidrol, X.; Balakirev, M.Y. High-Content Monitoring of Drug Effects in a 3D Spheroid Model. Front. Oncol. 2017, 7, 293.
- Gonzalez-Moragas, L.; Yu, S.M.; Murillo-Cremaes, N.; Laromaine, A.; Roig, A. Scale-up synthesis of iron oxide nanoparticles by microwave-assisted thermal decomposition. Chem. Eng. J. 2015, 281, 87–95.
- Gonzalez-Moragas, L.; Yu, S.M.; Benseny-Cases, N.; Stürzenbaum, S.; Roig, A.; Laromaine, A. Toxicogenomics of iron oxide nanoparticles in the nematode C. elegans. Nanotoxicology 2017, 11, 647–657.
- Stoica, A.-I.; Viñas, C.; Teixidor, F. Cobaltabisdicarbollide anion receptor for enantiomer-selective membrane electrodes. Chem. Commun. 2009, 33, 4988–4990.
- Stoica, A.-I.; Viñas, C.; Teixidor, F. Application of the cobaltabisdicarbollide anion to the development of ion selective PVC membrane electrodes for tuberculosis drug analysis. Chem. Commun. 2008, 48, 6492–6494.
- Stoica, A.-I.; Klebera, C.; Viñas, C.; Teixidor, F. Ion selective electrodes for protonable nitrogen containing analytes:Metallacarboranes as active membrane components. Electrochim. Acta 2013, 113, 94–98.
- Nuez-Martínez, M.; Pedrosa, L.; Martinez-Rovira, I.; Yousef, I.; Diao, D.; Teixidor, F.; Stanzani, E.; Martínez-Soler, F.; Tortosa, A.; Sierra, À.; et al. Synchrotron-based Fourier-transform infrared micro-spectroscopy (SR-FTIRM) fingerprint of the small anionic molecule Cobaltabisdicarbollide uptake in glioma stem cells. Int. J. Mol. Sci. 2021, 22, 9937, https://doi.org/10.3390/ijms22189937.
- Krenger, R.; Burri, J.T.; Lehnert, T.; Nelson, B.J.; Gijs, M.A.M. Force microscopy of the Caenorhabditis elegans embryonic eggshell. Microsyst. Nanoeng. 2020, 6, 29.
- Tarrés, M.; Viñas, C.; González-Cardoso, P.; Hänninen, M.M.; Sillanpää, R.; Dordovic, V.; Uchman, M.; Teixidor, F.; Matejicek, P. Aqueous self-assembly and cation selectivity of cobaltabisdicarbollide dianionic dumbbells. Chem. Eur. J. 2014, 20, 6786–6794.
- Fuentes, I.; García-Mendiola, T.; Sato, S.; Pita, M.; Nakamura, H.; Lorenzo, E.; Teixidor, F.; Marques, F.; Viñas, C. Metallacarboranes on the Road to Anticancer Therapies: Cellular Uptake, DNA Interaction, and Biological Evaluation of Cobaltabisdicarbollide [COSAN]− Chem. Eur. J. 2018, 24, 17239–17254.
- García-Mendiola, T.; Bayon-Pizarro, V.; Zaulet, A.; Fuentes, I.; Pariente, F.; Teixidor, F.; Viñas, C.; Lorenzo, E. Metallacarboranes as tunable redox potential electrochemical indicators for screening of gene mutation. Chem. Sci. 2016, 7, 5786–5797, doi:10.1039/c6sc01567k.
- Cigler, P.; Kožíšek, M.; Řezáčová, P.; Brynda, J.; Otwinowski, Z.; Pokorna, J.; Kožíšek, M.; Gruner, B.; Dolečková-Marešová, L.; Máša, M.; et al. From nonpeptide toward noncarbon protease inhibitors: Metallacarboranes as specific and potent inhibitors of HIV protease. Proc. Natl. Acad. Sci. USA 2005, 102, 15394–15399, doi:10.1073/pnas.0507577102.
- Fuentes, I.; Pujols, J.; Viñas, C.; Ventura, S.; Teixidor, F. Dual binding mode of metallacarborane produces a robust shield on proteins. Chem. A Eur. J. 2019, 25, 12820–12829, doi:10.1002/chem.201902796.
- Goszczynski, T.M.; Fink, K.; Kowalski, K.; Lesnikowski, Z.J.; Boratynski, J. Interactions of Boron Clusters and their Derivatives with Serum Albumin. Sci. Rep. 2017, 7, 9800.
- Evans, N.H.; Beer, P.D. Advances in Anion Supramolecular Chemistry: From Recognition to Chemical Applications. Angew. Chem. Int. Ed. 2014, 53, 11716–11754.
- Busschaert, N.; Caltagirone, C.; van Rossom, W.; Gale, P.A. Applications of Supramolecular Anion Recognition. Chem. Rev. 2015, 115, 8038–8055.
- Mathew, B.; Carradori, S; Guglielmi, P; Uddin, M.S.; Kim, H. New Aspects of Monoamine Oxidase B Inhibitors: The Key Role of Halogens to Open the Golden Door. Curr. Med. Chem. 2021, 28, 266–283.
- Alkorta, I.; Elguero, J.; Frontera, A. Not Only Hydrogen Bonds: Other Noncovalent Interactions. Crystals 2020, 10, 180.
- Borhani, S.; Mozdarani, H.; Babalui, S.; Bakhshandeh, M.; Nosrati, H. In vitro Radiosensitizing Effects of Temozolomide on U87MG Cell Lines of Human Glioblastoma Multiforme. Iran J. Med. Sci. 2017, 42, 258–265.





